Ankle Implants
SpiralUp TCL Allograft
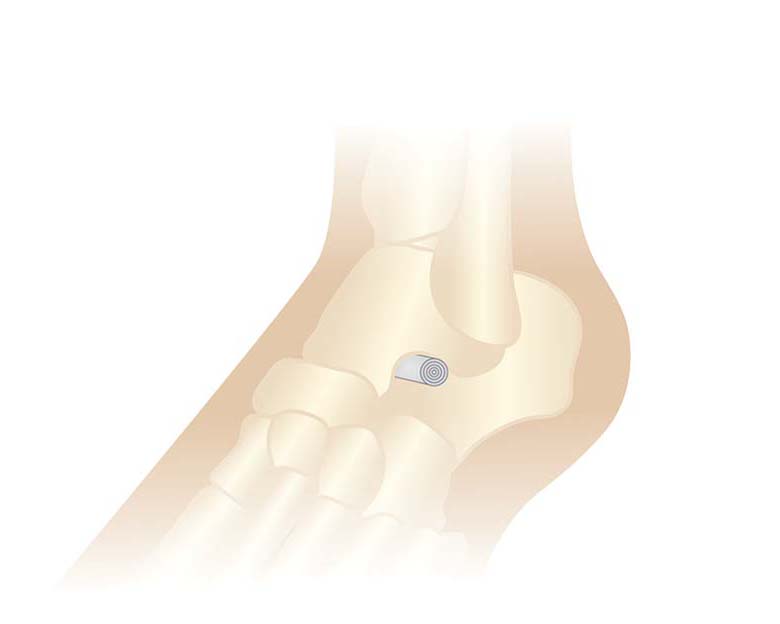
The SpiralUp® TCL Allograft System is an acellular dermal allograft intended to supplement the talocalcaneal ligament and as such, function as a dense, stable and spongey connective tissue layer. It is intended to improve the function of the foot, as well as conditions along the musculoskeletal (MSK) chain.
Talocalcaneal instability and peritalar subluxation are progressive conditions with far-reaching symptoms in a vast patient population. The SpiralUp TCL Allograft System is a unique approach to treating talocalcaneal instability and peritalar subluxation. The allograft is bullet shaped to support the anatomy of the canal and is engineered from acellular dermal tissue. It provides the necessary space in the sinus canal and also addresses ligamentous instability and peritalar subluxation. The SpiralUp implant is inserted across 80-90% of the sinus tarsi, it is able to stabilize the 3 facets (anterior, middle and posterior) of the subtalar joint more effectively than existing metal implants.
Key Features & Benefits:
- Pre-rolled, decellularized, freeze-dried, gamma sterilized human dermal allograft tissue
- Cannulated system
- Bullet shaped to support the anatomy of the tarsal canal
- Ease of implantation and removal
- No MRI restrictions
- Scaffold for tightening and integration of talocalcaneal ligaments
The SpiralUp TCL Allograft System is available in the US only.
The SpiralUp TCL Allograft System is not available outside of the United States.
Anika products may not be available in all geographies. Product availability is subject to the regulatory clearances in individual markets. Please reach out to your local representative or Contact Us if you have questions about specific market approvals.
For complete product information, including indications, contraindications, warnings, and precautions, please refer to the Instructions for Use found here.