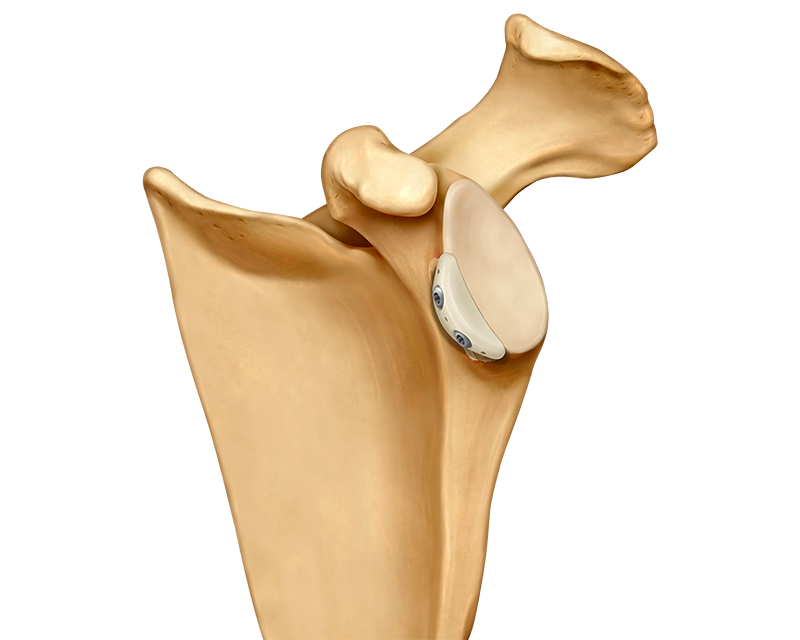
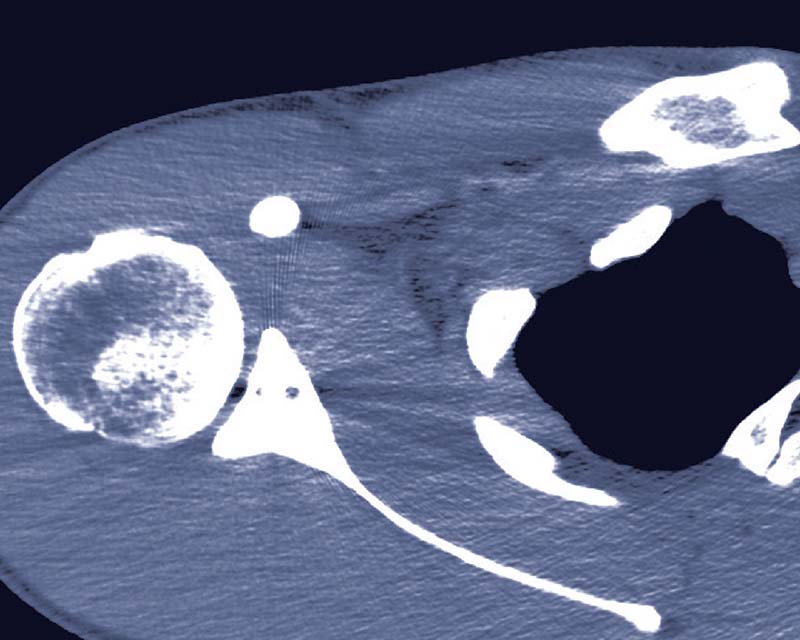
Glenojet Allograft System
Anika’s Glenojet® Allograft System was created to replace and augment anterior glenoid bone loss associated with trauma, recurrent dislocation or an unstable shoulder.
It is intended to be used for the repair, replacement, or reconstruction of musculoskeletal defects including bony pathologies associated with shoulder instability, such as anterior glenoid bone loss, bony Bankart, glenoid fracture or engaging Hill-Sachs lesions.
The Glenojet is designed to match the contour of the glenoid defect and eliminates the procedural steps and surgical time associated with autograft bone harvesting and preparation and is a great alternative to a traditional latarjet procedure.1
Key Features & Benefits:
- Pre-shaped, pre-drilled human cortical allograft
- Pre-drilled holes eliminate the risk of preparation fractures and improve suture management
- Suture holes allow intra or extra-articular soft tissue fixation
- Use of allograft avoids:
- Donor site irritation for autograft bone block1
- Extensive dissection and risk of neurovascular injury from coracoid transfer1
- Sling effect can still be achieved without the need for dissecting the coracoid
- Excellent revision option for failed latarjet
- Single-use, disposable and sterile instruments offer a reproducible technique
- Glenoid reamers create a congruent surface to optimize glenoid to graft interface while creating a precise and reproducible cut plane to expedite glenoid preparation
Anika products may not be available in all geographies. Product availability is subject to the regulatory clearances in individual markets. Please reach out to your local representative or Contact Us if you have questions about specific market approvals.
For complete product information, including indications, contraindications, warnings, and precautions, please refer to the Instructions for Use found here.
- Smucny, M. and Miniaci, A. Pre-Shaped Allograft for Glenoid Reconstruction in Anterior Shoulder Instability. Arthroscopy Techniques, Vol 7, No 4 (April), 2018: pp e343-e348