Harnessing the power of hyaluronic acid for regenerative solutions and osteoarthritis pain management
At Anika, we harness the power of hyaluronic acid (HA) to develop innovative solutions for orthopedic conditions. We are recognized worldwide as industry pioneers with more than 30 years of expertise in developing, manufacturing, and commercializing HA-based products. Our proprietary HA technology platform is built on decades of research, clinical validation, and a commitment to improving patient lives.
What is Hyaluronic Acid
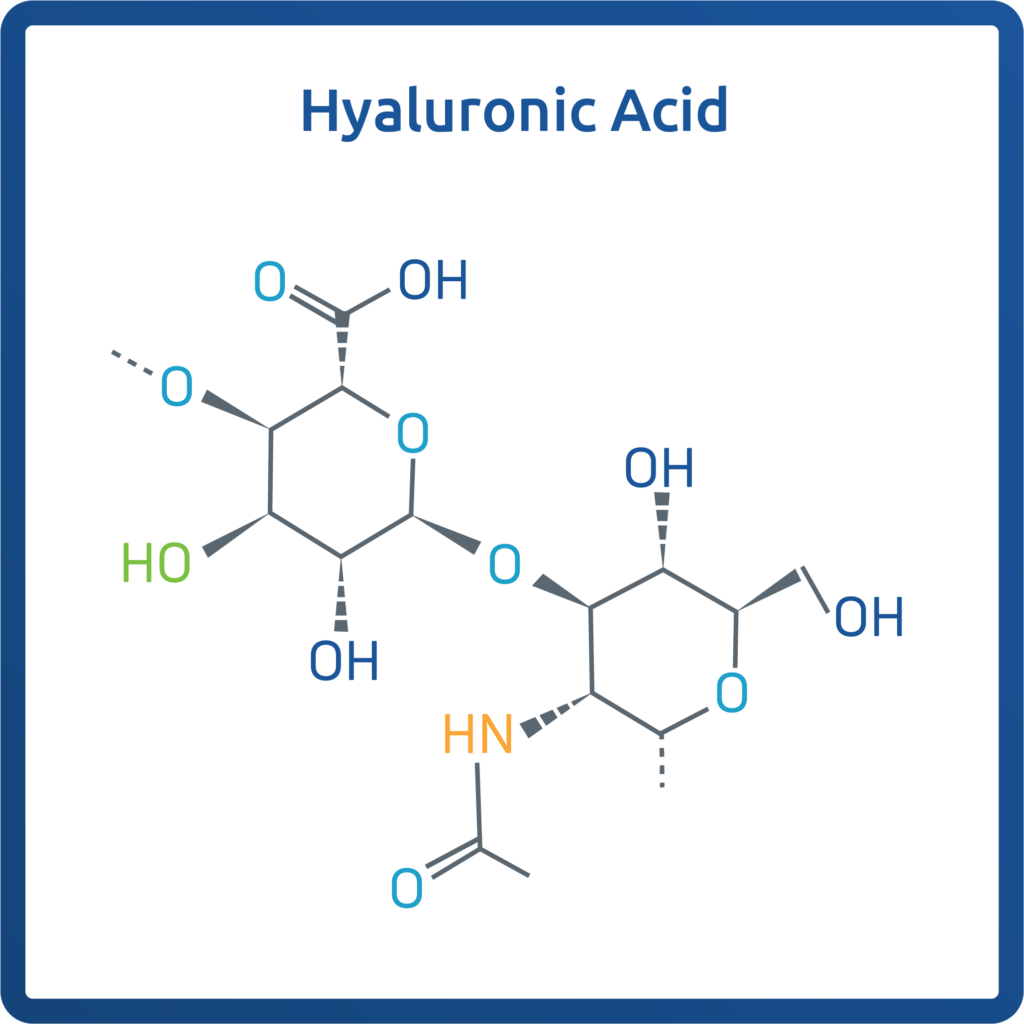
Hyaluronic acid (HA) is a naturally occurring substance found throughout the human body, with high concentrations in the extracellular matrix (ECM) of connective tissues, skin, eyes, and synovial fluid found within joints. In the joints, HA plays a vital role in lubrication, shock absorption, and hydration, contributing to the biomechanical integrity and homeostasis (stability) of articular cartilage.
Beyond its mechanical functions of cushioning and lubricating, HA is a key structural component of the ECM by supporting cell migration, proliferation, and differentiation, helping cells move to the injury site, multiply, and specialize into the right tissue type while also regulating inflammatory responses.1 Its viscoelastic (solid and liquid properties) and hydrophilic (water-liking) properties make it essential for maintaining tissue architecture and promoting healing. In simple terms, HA acts as a natural lubricant and plays a vital role in tissue health and repair.2
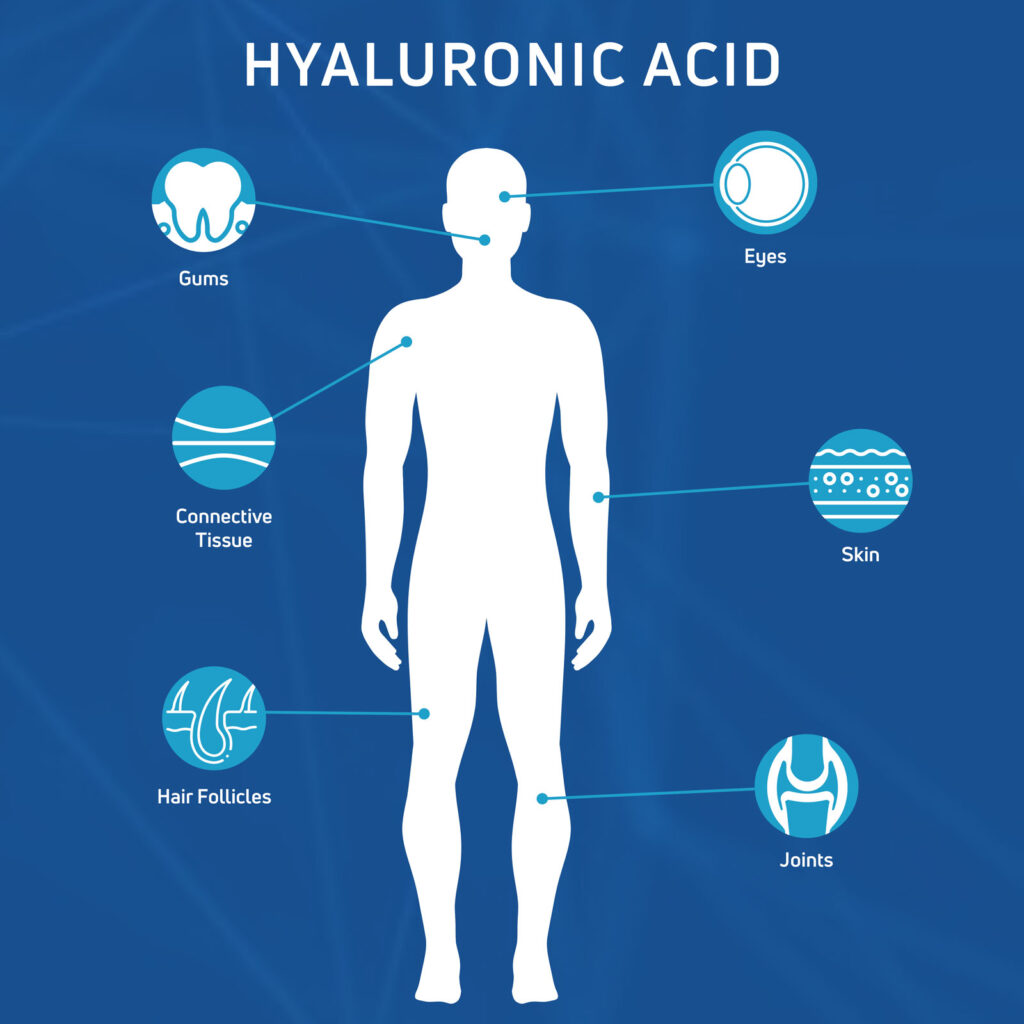
We refine and formulate bio-fermented, non-avian (non-animal) sourced HA to enhance its therapeutic potential across a range of orthopedic and regenerative applications. Our proprietary HA technologies, including Hyaff® (esterified hyaluronic acid) and cross-linked HA, extend the residence time of HA (i.e. the amount of time HA stays in the body) and enable its use in injectable therapies, implantable scaffolds, and combination biologic solutions.
Our HA-based products are designed to reduce pain, restore joint function, and support tissue regeneration. By leveraging HA’s natural biology and engineering it for clinical performance, we help patients regain mobility and quality of life.
Hyaff-11 (Hyaff): Our Proprietary Regenerative Hyaluronic Acid Technology
Hyaff is 100% solid esterified hyaluronic acid (HA), our proprietary technology that was developed to overcome the limitations of native HA in medical applications. Unlike native HA, which degrades rapidly, Hyaff has a longer residence time (lasts longer in the body), allowing for sustained therapeutic effects. Hyaff is a solid phase material that can be engineered into highly porous biodegradable scaffolds that support cellular infiltration and tissue regeneration. Therefore, the Hyaff fibers are essentially an HA-based regenerative material that stays in the body long enough to support and promote tissue healing before it resorbs.
Composition and Properties
Hyaff is modified through the esterification of HA with benzyl alcohol, making it water-insoluble and resulting in porous 100% hyaluronic acid-based solid materials such as:
- Non-woven scaffolds (similar to gauze)
- Knitted scaffolds (similar to a knitted hat)

Mechanism of Action
As Hyaff degrades, it gradually releases HA, creating a localized HA-rich environment that supports:
It mimics the embryonic extracellular matrix, promoting scarless repair and mesenchymal stem cell (MSC) recruitment, especially in tendon healing and cartilage repair.4,5


Clinical Applications
Used in products like Hyalofast® and Integrity™, Hyaff supports:
- Chondral and osteochondral (cartilage) lesion repair
- Rotator cuff, Achilles, and other tendon augmentation procedures
Hyaff has demonstrated a strong safety and efficacy profile in over 20 years of global clinical use.8

Cross-linked Hyaluronic Acid Gel: Our Proprietary Injectable Technology for Osteoarthritis Pain Management
Cross-linked hyaluronic acid (HA) is engineered to overcome the limitations of native HA in managing osteoarthritis pain. Unlike native HA, which breaks down quickly in the joint, cross-linked HA is chemically stabilized to last longer, providing extended lubrication and cushioning for sustained relief.
Through our proprietary cross-linking process, HA chains are modified to create a more durable material with enhanced mechanical properties that remains in the body for an extended period. This transformation turns HA from a liquid into a viscoelastic gel that remains in the joint space longer, helping reduce pain and improve joint function.
Our Non-Orthopedic Solutions
In addition to our primary focus around regenerative solutions and osteoarthritis pain management, our hyaluronic acid-based portfolio also includes non-core products in the areas of:
-
- Animal Health
- Ophthalmic
- Other non-orthopedic solutions
Technology Leadership
Built on decades of hyaluronic acid (HA) innovation and clinical validation
30+ Years of Expertise
Industry pioneers with decades of experience in developing, manufacturing, and commercializing HA-based solutions
Proprietary Technology
Advanced HA technology platform including linear HA, cross-linked HA, and Hyaff for enhanced therapeutic performance
Orthopedic Solutions are Non-Animal Sourced
Our regenerative solutions and osteoarthritis (OA) pain management injections are all naturally derived forms of hyaluronic acid from non-avian sourced HA
Clinically Validated
Decades of research and clinical validation supporting product safety and efficacy
Global Reach
Products available worldwide in 45+ countries
Continuous Innovation
Ongoing commitment to advancing regenerative medicine and improving patient outcomes
- Polizzi, A., et al. Impact of hyaluronic acid and other Re-Epithelializing agents in periodontal regeneration: A molecular perspective. Int. J. Mol. Sci. 25.22 (2024): 12347. https://doi.org/10.3390/ijms252212347(opens in a new tab)
- Regeneration: A Molecular Perspective. Int. J. Mol. Sci. 2024, 25, 12347. https://doi.org/10.3390/ijms252212347(opens in a new tab)
- Marinho, Andreia, et al. “Hyaluronic acid: a key ingredient in the therapy of inflammation.” Biomolecules 11.10 (2021): 1518.
- Pasquinelli G, Orrico C, Foroni L, Bonafè F, Carboni M, Guarnieri C, Raimondo S, Penna C, Geuna S, Pagliaro P, Freyrie A, Stella A, Caldarera CM, Muscari C. Mesenchymal stem cell interaction with a non-woven hyaluronan-based scaffold suitable for tissue repair. J Anat. 2008 Nov;213(5):520-30. doi: 10.1111/j.1469-7580.2008.00974.x. PMID: 19014359; PMCID: PMC2667546. https://pubmed.ncbi.nlm.nih.gov/19014359/(opens in a new tab)
- Longaker, M. T. et al. Studies in fetal wound healing, VII. Fetal wound healing may be modulated by hyaluronic acid stimulating activity in amniotic fluid. J. Pediatr. Surg. 25, 430–433 (1990).
- Hu, Min, et al. “Three dimensional hyaluronic acid grafts promote healing and reduce scar formation in skin incision wounds.” J. Biomed. Mater. Res., 67.1 (2003): 586-592
- Milella, E., et al. “Physico-chemical properties and degradability of non-woven hyaluronan benzylic esters as tissue engineering scaffolds.” Biomaterials. 23.4 (2002): 1053-1063.
- Vindigni, V. Hyaluronan Benzyl Ester as a Scaffold for Tissue Engineering. Int. J. Mol. Sci. 2009, 10, 2972-2985; doi:10.3390/ijms10072972
- Data on file, Anika Therapeutics, Inc.
