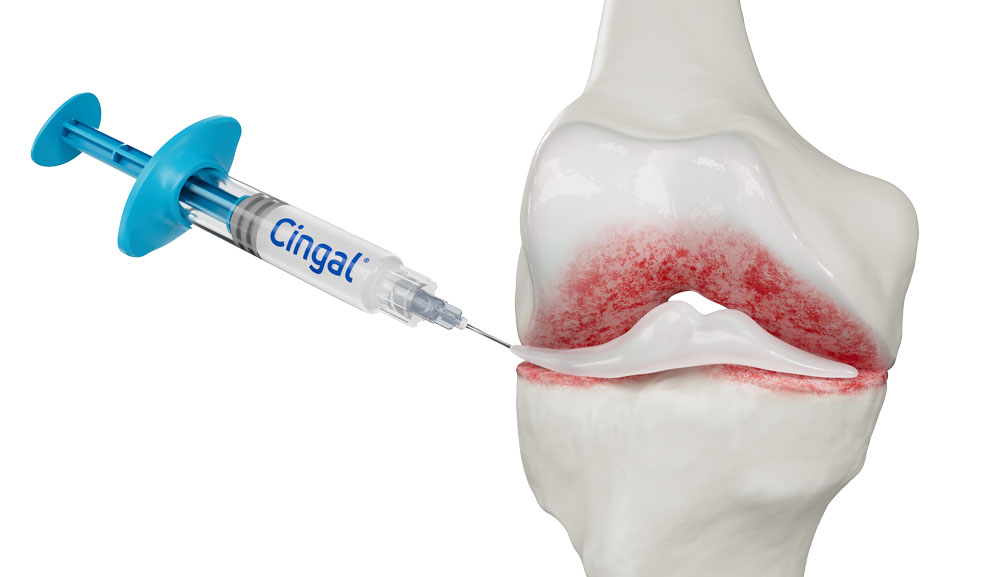
What Is Cingal?
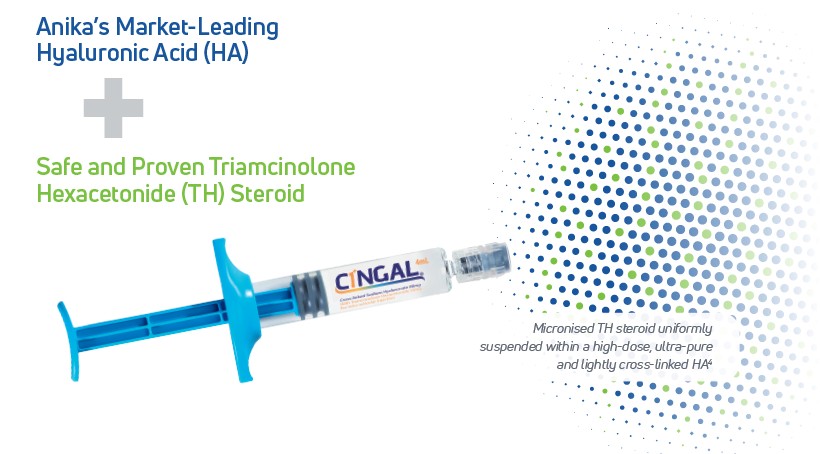
Cingal® is a next generation, single injection treatment proven to safely and effectively treat the pain, stiffness and functions associated with osteoarthritis (OA)1. Cingal combines two well-established treatments into a single injection, providing the benefits of both an approved corticosteroid, triamcinolone hexacetonide (TH), and our proprietary high concentration cross-linked hyaluronic acid formulation (Monovisc).
The corticosteroid reduces inflammation in the knee joint, providing rapid pain relief within days of injection, while the hyaluronic acid component works in parallel to deliver long-lasting pain relief proven to last through 6 months.1 Cingal is a simple treatment that is administered by a healthcare professional during a routine office visit.
Real Life Advantages
Next generation, non-opioid OA pain management: First and only approved* combination hyaluronic acid (HA) and corticosteroid for osteoarthritis pain
Clinically Proven Fast-Acting & Long-Lasting Pain Relief
- Fast-acting pain relief: Triamcinolone hexacetonide (TH) treats inflammation and provides rapid pain relief within days of injection1
- Long-lasting pain relief: HA provides sustained pain relief through 6 months1
- Clinically proven: Unmatched combination of both rapid and long-lasting knee pain relief across three Phase III clinical trials1,2,3
- Safe and effective: Strong safety profile in both an initial and repeat injection1,4
Advanced Hyaluronic Acid (HA) Formulation
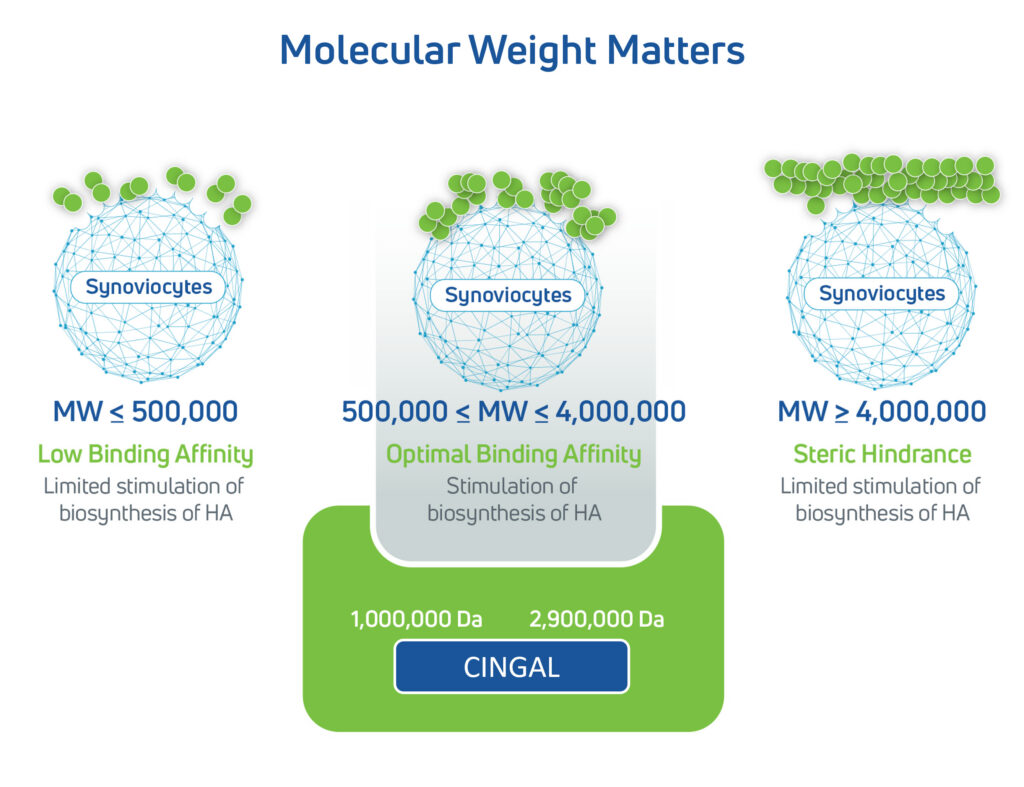
- Natural solution: Highly concentrated, non-animal-based HA produced from bacterial fermentation
- High HA concentration: 22 mg/mL, one of the highest available in its class
- Proprietary HA formulation: Enhances the synovial fluid’s viscoelasticity, improves mobility, cushions joints, and protects articular cartilage
- Biocompatible and resorbable: Safe and naturally broken down by the body
Convenience & Availability
- Convenient single-injection treatment: Simple injection administered during a routine office visit with no need for multiple visits
- Global availability: Registered and approved in over 45 countries worldwide
How It Works
All joints in the body contain synovial fluid, a vital substance that provides lubrication, cushioning, and protection as we move. This fluid is naturally rich in hyaluronic acid (HA), which helps reduce friction and absorb shock during joint activity.
Over time, especially in patients with osteoarthritis (OA), the synovial fluid becomes degraded, losing both its quality and volume. This leads to increased joint friction, inflammation, stiffness, and pain. Cingal addresses this by combining two proven therapies in a simple injection that may be done in the convenience of a clinician’s office:
HA viscosupplement
Replenishes lost HA to restore the fluid’s natural viscosity and cushioning properties. This improves joint mobility and delivers long-term pain relief, lasting through 6 months1.
Triamcinolone hexacetonide (TH)
A powerful corticosteroid that reduces inflammation and provides rapid pain relief within days. TH remains active in the joint for up to 21 days, longer than typical steroids7
Together, HA and TH work synergistically
The actions of Cingal are long-term relief of symptoms by lubrication and mechanical support supplemented by short-term pain relief provided by triamcinolone hexacetonide.8
Evidence
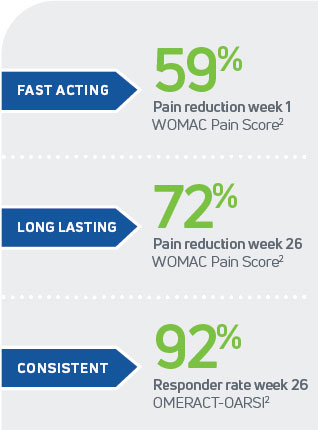
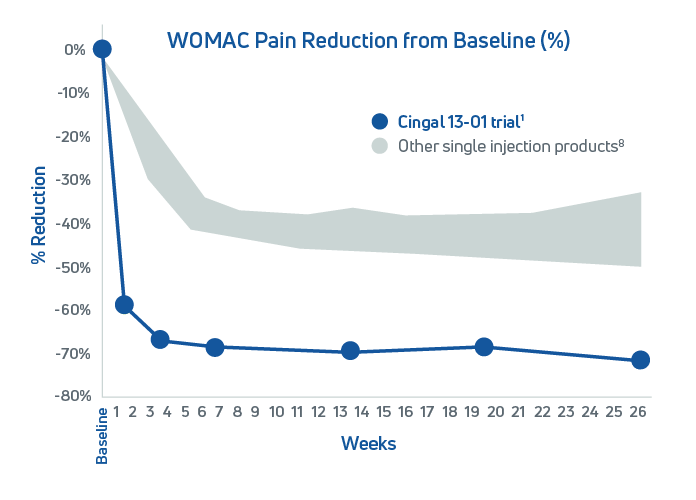
Cingal is backed by robust data from three Phase III clinical trials, Cingal 13-01, 16-02, and 19-011, 2, 3 In the pivotal Cingal 13-01 study, a randomized, double-blind, placebo-controlled, multi-center trial involving 368 patients with knee osteoarthritis (Kellgren-Lawrence grades I–III), Cingal consistently outperformed both saline placebo and a leading single-injection HA only product.1 The results speak for themselves:
- Statistically significant improvements in pain, stiffness, physical function, and global assessments
- Rapid pain relief within the first week
- Sustained symptom improvement through 26 weeks
- OMERACT-OARSI responder rates of 89% at Week 1 and 92% through Week 26 confirm its strong clinical impact
In the Cingal 19-01 clinical study, Cingal demonstrated superiority over triamcinolone hexacetonide (TH) steroid alone for patients suffering with osteoarthritic knee pain at 26 weeks.3 Together over its three Phase III clinical trials, Cingal has demonstrated superiority over each of its active ingredients and placebo.1,2,3
A follow-up retreatment study (Cingal 13-02) confirmed the safety of repeat injections.4 In addition to its strong efficacy, Cingal has a well-established safety profile supported by extensive clinical data demonstrating its biocompatibility and tolerability. This makes it a trusted choice for physicians and patients seeking long-lasting osteoarthritis symptom relief without compromising safety.
“The pivotal study results unequivocally demonstrate the strong clinical benefit of Cingal. Patients receiving Cingal experienced significant symptom relief from the first week that continued and even strengthened throughout six months.”
These results highlight Cingal’s unique dual-action formulation, combining fast-acting corticosteroid relief with long-term mechanical support from cross-linked hyaluronic acid (HA), and reinforce its role as a powerful, non-surgical treatment option for knee osteoarthritis.
Since its launch, over one million Cingal injections have been performed worldwide.9 This reflects its growing global adoption and clinical value in delivering both rapid and sustained symptom relief for patients with OA.
Select Publications & Clinical Data
For a full list of publications, please Contact Us.
Laszlo Hangody, Robert Szody, Piotr Lukasik, Wojciech Zgadzaj, Endre Lénárt, Eva Dokoupilova, Daniela Bichovsk, Agnes Berta, Gabor Vasarhelyi, Andrea Ficzere, György Hangody, Gary Stevens, Miklos Szendroi
Key Takeaways
- Randomized, double-blind, placebo-controlled multicenter trial evaluating Cingal (cross-linked sodium hyaluronate + triamcinolone hexacetonide) for knee osteoarthritis
- Patients (n=368) received either Cingal, hyaluronic acid alone, or saline placebo.
- Cingal group showed significantly greater pain relief at early time points (weeks 1–3) compared to both comparators.
- Sustained improvement in WOMAC scores through 26 weeks, especially in pain and function domains.
- No serious adverse events reported; safety profile comparable across all groups.
- Conclusion: Cingal provides rapid and sustained symptom relief in knee OA, combining the benefits of HA and corticosteroid in a single injection.
Etienne L Belzile, Robert T Deakon, Christopher Vannabouathong, Mohit Bhandari, Martin Lamontagne and Robert McCormack
Key Takeaways
- Cost-utility analysis comparing a single-injection formulation of corticosteroid + hyaluronic acid (Cingal) vs. a 2-injection regimen of sequential corticosteroid and HA.
- Single-injection Cingal was found to be cost-effective, assuming a willingness-to-pay threshold of $50,000 per quality-adjusted life year (QALY) gained.
- Economic modeling showed lower overall treatment costs and comparable sustained symptom relief with the single-injection approach.
- Simplified administration and reduced patient burden make Cingal a favorable option in clinical practice.
- Conclusion: Cingal offers a cost-effective and efficient alternative to multi-injection regimens for knee OA symptom management.
Christoph Bauer, Lukas B. Moser, Vivek Jeyakumar, Eugenia Niculescu-Morzsa, Daniela Kern, Stefan Nehrer
Key Takeaways
- In vitro study using human osteoarthritic chondrocytes treated with inflammatory cytokines (IL-1β and IL-17) to simulate OA conditions.
- Compared effects of glucocorticoid (triamcinolone hexacetonide), hyaluronic acid (HA), and their combination on chondrocyte health and inflammation.
- Combined GC/HA treatment enhanced metabolic activity, reduced catabolic gene expression, and lowered TNF-α release more effectively than either agent alone.
- HA mitigated the negative cellular effects of glucocorticoids, such as reduced actin polymerization and increased oxidative stress.
- Conclusion: Co-administration of HA and GC offers superior anti-inflammatory and chondroprotective effects compared to separate administration, supporting the rationale for combination therapies in OA management.
Resources
Product Information
Access comprehensive technical documentation and marketing materials
- Brochure
- Injection Technique
Instructions for Use
Complete product specifications and safety information
In the European Economic Area and other parts of the world, Cingal is indicated as a viscoelastic supplement or a replacement for synovial fluid in human knee joints. Cingal is well suited for rapid and long-term relief of the symptoms of human knee joint dysfunctions, such as osteoarthritis. The actions of Cingal are long-term relief of symptoms by lubrication and mechanical support supplemented by short-term pain relief provided by triamcinolone hexacetonide.8
In Canada, Cingal is indicated for the treatment of pain in osteoarthritis (OA) of the knee in patients who have failed to respond adequately to conservative non-pharmacologic therapy and to simple analgesics (e.g. acetaminophen). Cingal includes an ancillary steroid to provide additional short-term pain relief.10
Cingal is not yet available in the United States, but progress is actively underway. Anika is engaged in ongoing dialogue with the FDA and is making steady progress toward an Investigational New Drug (IND) submission. This important regulatory step reflects Anika’s commitment to bringing Cingal to U.S. patients, where its proven clinical efficacy and safety profile have already earned widespread adoption internationally.
Anika products may not be available in all geographies. Product availability is subject to the regulatory clearances in individual markets. Please reach out to your local representative or Contact Us if you have questions about specific market approvals.
For complete product information, including indications, contraindications, warnings, and precautions, please refer to the Instructions for Use found here(opens in a new tab).
- *Cingal is CE Mark approved and Health Canada approved. Cingal is not approved for use in the USA.
- Hangody L, et al. Intraarticular injection of a cross-linked sodium hyaluronate combined with triamcinolone hexacetonide (Cingal) to provide symptomatic relief of osteoarthritis of the knee: a randomized, double-blind, placebo-controlled multicenter clinical trial. Cartilage. 2018 Jul;9(3):276-283. doi: 10.1177/1947603517703732. Epub 2017 May 23. PMID: 28535076; PMCID: PMC6042027. https://pmc.ncbi.nlm.nih.gov/articles/PMC6042027/(opens in a new tab)
- 16-02 Data on File
- 19-01 Data on File
- 13-02 Data on File
- Smith MM, Ghosh P. The synthesis of hyaluronic acid by human synovial fibroblasts is influenced by the nature of the hyaluronate in the extracellular environment. Rheumatol Int. 1987; 7(3):113-22 https://pubmed.ncbi.nlm.nih.gov/3671989/(opens in a new tab)
- R009-041, Technical Report, “Chemical Stability Evaluation of Cross-Linked Hyaluronic Acid and Triamcinolone Hexacetonide in Cingal Product”, December 2009. Appendix II of the device dossier.
- Stephens M, et al. Musculoskeletal injections: a review of the evidence. Am Fam Physician. 2008 Oct 15;78(8):971-976. https://www.aafp.org/pubs/afp/issues/2008/1015/p971.html(opens in a new tab)
- AML-500-306/D 08-2020 IFU
- Data on file, Anika Therapeutics, Inc.
- AML-500-305/B IFU