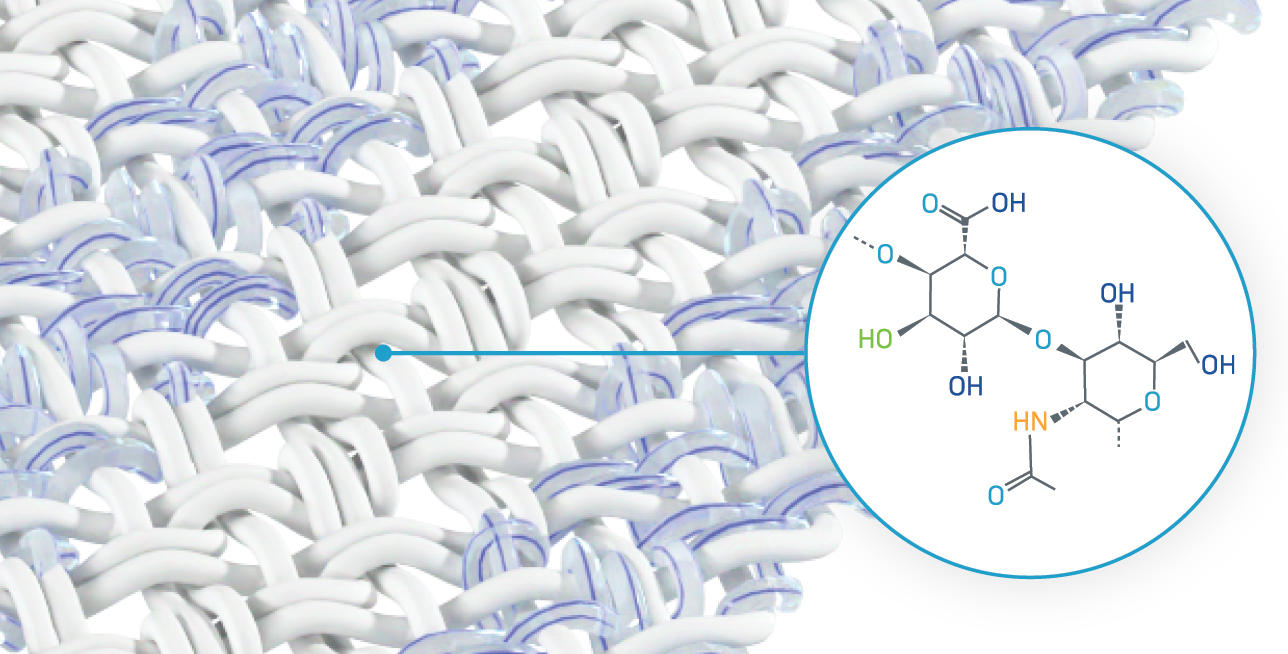
Hyaluronic acid (HA) is essential for maintaining healthy joints and tissue. It offers a unique advantage in healing, including regulating inflammation, supporting cell infiltration and differentiation, and helping foster an environment for healthy tissue repair.1-4 In the joints, hyaluronic acid acts as a lubricant and shock absorber, which is pivotal in fostering pain-free range of motion.1,5 With these important capabilities, it’s only natural to use HA when creating products to support joint health.
However, HA is viscous (thick, sticky consistency) and breaks down rapidly in the body.6 To make it more usable for medical purposes, scientists needed to determine a way to make HA last longer in the body.
Anika’s scientists are able to chemically modify HA to change it from a liquid gel form and manufacture it into a solid form, making a more versatile derivative of HA, Hyaff-11. This remarkable breakthrough in medical science provides an opportunity for applications within the body.
What is Hyaff
Hyaff-11 is a chemically modified form of biodegradable hyaluronic acid. This chemical modification (esterification with benzyl alcohol) means:
- Hyaff is water-insoluble, meaning it can be made into many different solid-form configurations including non-woven scaffolds of Hyaff fibers, sponges, granules, and membranes7,8
- Hyaff lasts longer in the body than native HA (increased residence time) to support the healing process9
- As Hyaff degrades, it releases HA, providing a unique (and natural) HA-enriched environment at the implant site7
Is Hyaff clinically proven?
Hyaff has been used globally for more than 20 years, with excellent safety and efficacy results.10 Clinical studies have demonstrated that as Hyaff fibers degrade, hyaluronic acid is released, creating an HA-rich embryonic-like environment favorable to tissue regeneration and healing.11 Furthermore, Anika has over 30 years (since 1992) of experience developing, manufacturing, and selling HA-based products for a variety of anatomies and applications.
What is Anika’s most recent Hyaff-based product?
Anika’s recently launched Integrity Implant System is at the forefront of rotator cuff repair. Integrity is designed to protect injured tendons and support regenerative healing in rotator cuff and other tendon repairs. The implant is based on Anika’s proprietary Hyaff hyaluronic acid technology and is reinforced with PET (polyethylene terephthalate), a material known for its biostability, support of tissue growth, and high strength.12
Anika continues to invest in research and development to create hyaluronic acid-based solutions that help patients maintain an active lifestyle. To learn more about Anika, Hyaff, its history, or future, please visit our website: https://anika.com/
References:
- Necas, J. B. L. B. P., et al. “Hyaluronic acid (hyaluronan): a review.” Veterinarni medicina 53.8 (2008): 397-411.
- Spicer, Andrew P., and Janet YL Tien. “Hyaluronan and morphogenesis.” Birth Defects Research Part C: Embryo Today: Reviews. 72.1 (2004): 89-108.
- Zhang, Ling-Tao, et al. “Hyaluronic acid promotes osteogenic differentiation of human amniotic mesenchymal stem cells via the TGF-β/Smad signalling pathway.” Life Sciences. 232 (2019): 116669
- Cooper, Carol A., et al. “Inflammation and hyaluronic acid.” Alternative & complementary Therapies. 14.2 (2008): 78-84.
- Swann, D. A. et al. Role of hyaluronic acid in joint lubrication. Ann. Rheum. Dis. 33, 318–326 (1974).
- Laurent, U. B. G. & Reed, R. K. Turnover of hyaluronan in the tissues. Adv. Drug Deliv. Rev. 7, 237–256 (1991).
- Milella, E., et al. “Physico-chemical properties and degradability of non-woven hyaluronan benzylic esters as tissue engineering scaffolds.” Biomaterials. 23.4 (2002): 1053-1063.
- Vindigni, V. Hyaluronan Benzyl Ester as a Scaffold for Tissue Engineering. Int. J. Mol. Sci. 2009, 10, 2972-2985; doi:10.3390/ijms10072972
- Turner, N. et al. A novel hyaluronan-based biomaterial (Hyaff-11s) as a scaffold for endothelial cells in tissue engineered vascular grafts. Biomaterials, 25 (2004) 5955-5964
- Data on file. Publications available upon request.
- Longaker, M. T. et al. Studies in fetal wound healing, VII. Fetal wound healing may be modulated by hyaluronic acid stimulating activity in amniotic fluid. J. Pediatr. Surg. 25, 430–433 (1990).
- Metzger, A. Polyethylene Terephthalate and the Pillar™ Palatal Implant: Its Historical Usage and Durability in Medical Applications. Restore Medical. 2003.