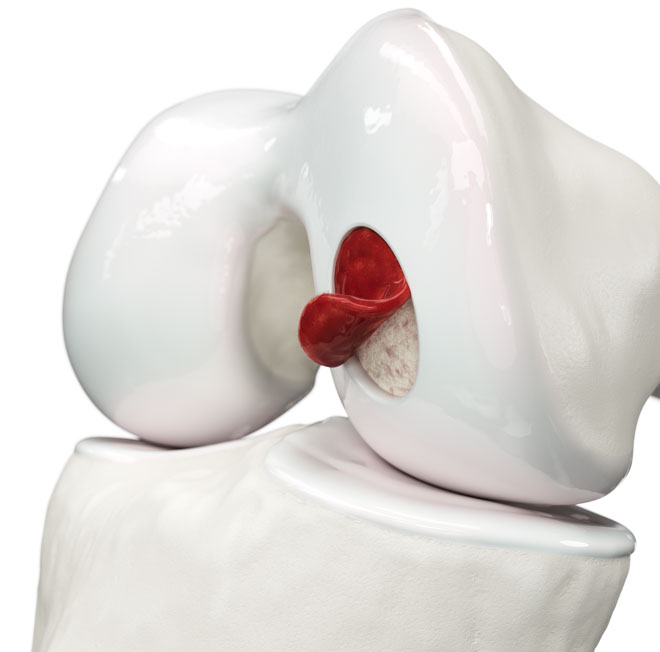
What Is Hyalofast?
Hyalofast is a biodegradable implant designed to support the repair of cartilage injuries. Hyalofast is made entirely from Hyaff®, a chemically modified form of hyaluronic acid (a benzyl ester of hyaluronic acid) and is designed to entrap stem cells, including mesenchymal stem cells (MSCs) from bone marrow aspirate concentrate (BMAC) for the repair of cartilage defects, specifically chondral and osteochondral lesions.1,2 It can also act as a chondroprotective layer, helping retain MSCs at the site following bone marrow stimulation (BMS) procedures.
The scaffold’s fibrous structure promotes cell adhesion, proliferation, and differentiation, allowing MSCs to organize within its three-dimensional structure to support the creation of functional cartilage.3 As it gradually degrades, it enriches the site with hyaluronic acid, a key component of the extracellular matrix, and supports cartilage regeneration.1,4
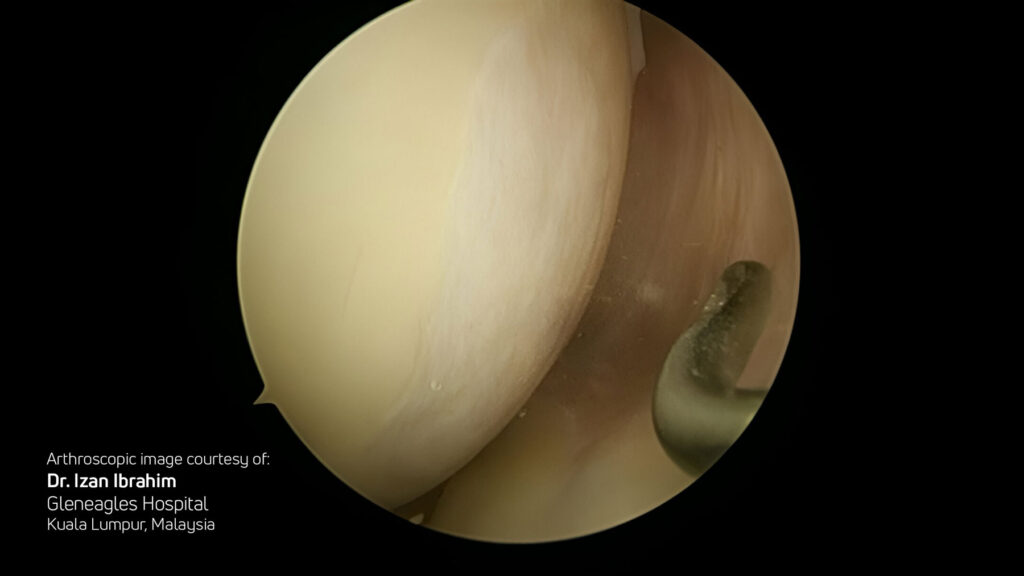
Hyalofast is off-the-shelf, easy to handle, and can be implanted via arthroscopy or mini-arthrotomy in a single surgery procedure. Its soft, conformable texture allows it to adapt to the shape of the lesion and adhere to the site without the need for additional fixation in most cases. Its uniform single-layer 3D structure allows application in any orientation, including stacking if needed.
Real Life Advantages
Hyalofast enhances the body’s cellular response and supports the natural cartilage healing process.

Hyalofast has two roles:
- Structural: 3D structure of Hyalofast facilitates the entrapment of mesenchymal stem cells (MSCs) and cell adhesion, supporting their multi-dimensional development3
- Biologic: As Hyalofast degrades, it creates an HA-rich environment that favors chondrogenesis (the biological process of cartilage formation)4,1

Simple, Single-Stage, Versatile
- Specifically designed for simple and easy implantation
- Single-stage (one surgery), off-the-shelf, arthroscopic or mini-open techniques
Clinically Proven
- Second look biopsy histology, and T2 mapping confirm durable hyaline-like cartilage7,8,9,10,11
- Backed by up to 16 years of data showing significant improvements in pain and function12
Evidence
Clinical studies have shown Hyalofast to be safe and effective for treating cartilage lesions in the ankle and knee.12 It supports the formation of hyaline-like cartilage, integrates smoothly with surrounding tissue, and leads to pain reduction and functional improvement, enabling patients to return to daily activities and non-impact sports with high satisfaction.7-12
With data out to 16 years, Hyalofast is a clinically proven solution for cartilage repair. Hyalofast has demonstrated positive clinical results in:
Short-to Medium-Term Outcomes (up to 8 years):
- Buda et al. (2010, 2013): Demonstrated successful one-stage cartilage repair using Hyalofast with BMAC7
- Vannini et al. (2012): Showed positive outcomes using T2 mapping in patients with juvenile osteochondritis dissecans9
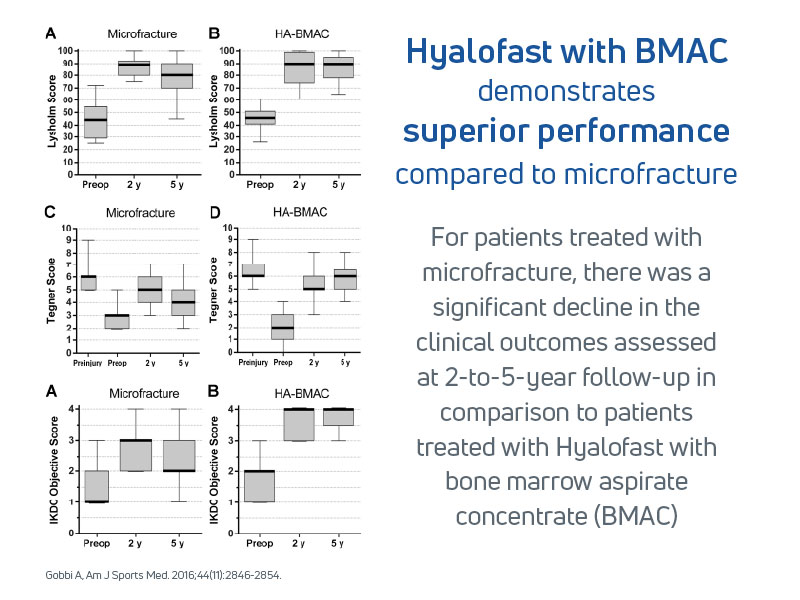
- Gobbi et al. (2016): Compared Hyalofast loaded with BMAC to microfracture alone, showing superior clinical results at 5-year follow-up13
Comparing Effectiveness Across Treatment Options:
- Sofu et al. (2017): Found that Hyalofast combined with microfracture (MFX) yielded statistically significant improvements in clinical and MRI outcomes compared to MFX alone14
- Gobbi et al (2019): Hyalofast single-stage surgery clinical results were equivalent to the matrix-induced autologous chondrocyte implantation (MACI) technique, which requires 2 surgeries, at medium-term follow-up10

Clinical Applications in Specific Patient Groups:
- Whyte et al. (2024) Hyalofast was effective in large patellofemoral chondral defects, in patients over 45 years old with realignment procedures12
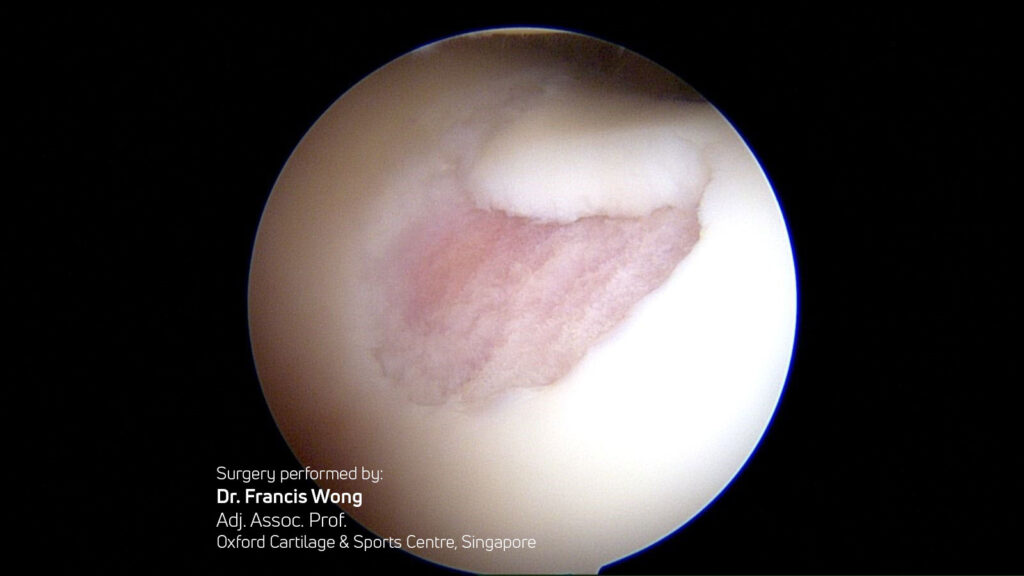
Quality of Regenerated Tissue:
- Buda, Vannini, Gobbi (multiple) Hyaline-like cartilage regeneration confirmed via second-look biopsies, arthroscopies, and MRI T2 mapping.7-11
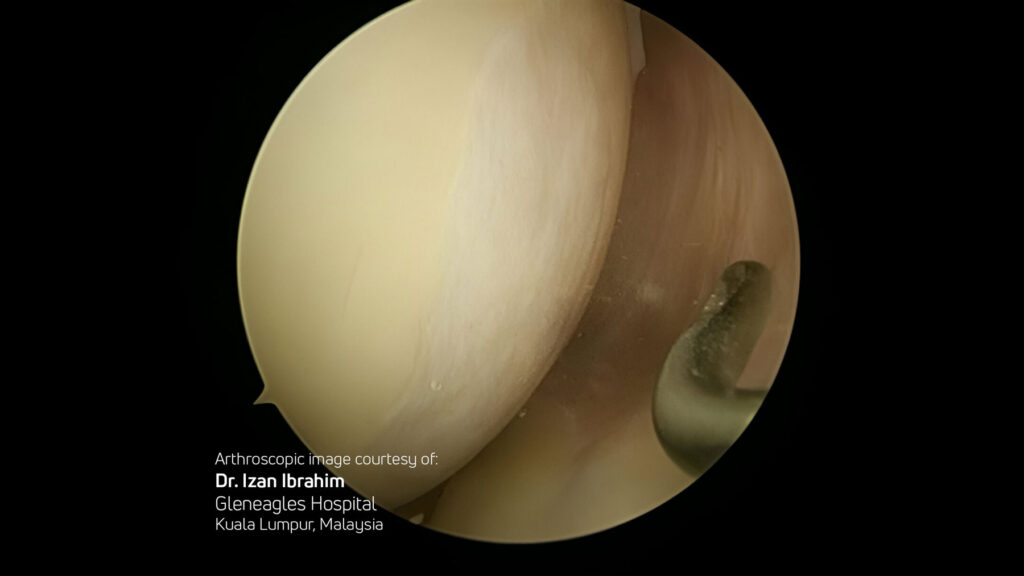
Select Publications & Clinical Data
For a full list of publications, please Contact Us.
(Roberto Buda, Francesca Vannini, Marco Cavallo, Brunella Grigolo, Annarita Cenacchi, Sandro Giannini)
Key Takeaways
- Prospective study of 48 patients with femoral condyle lesions treated using Hyalofast with bone marrow concentrate
- Significant improvements in pain and function scores at 6 and 12 months
- MRI confirmed scaffold integration and defect filling
- Most patients returned to activity within 6–12 months; no major complications reported
(Francesca Vannini , Milva Battaglia, Roberto Buda, Marco Cavallo, Sandro Giannini)
Key Takeaways
- 28 patients (mean age 15.2) with stable JOCD lesions treated with Hyalofast and bone marrow concentrate
- Clinical scores improved significantly at 6 and 12 months
- T2 mapping showed progressive cartilage maturation
- Safe, effective, and minimally invasive for young patients
(Alberto Gobbi , Graeme P Whyte )
Key Takeaways
- 50 patients randomized to Hyalofast with BMAC arm or microfracture arm
- Scaffold group showed superior clinical scores and MRI outcomes at 1–2 years
- Sustained improvements at 5 years; microfracture group results declined
- Faster return to sport and no major complications
(Alberto Gobbi , Graeme P Whyte )
Key Takeaways
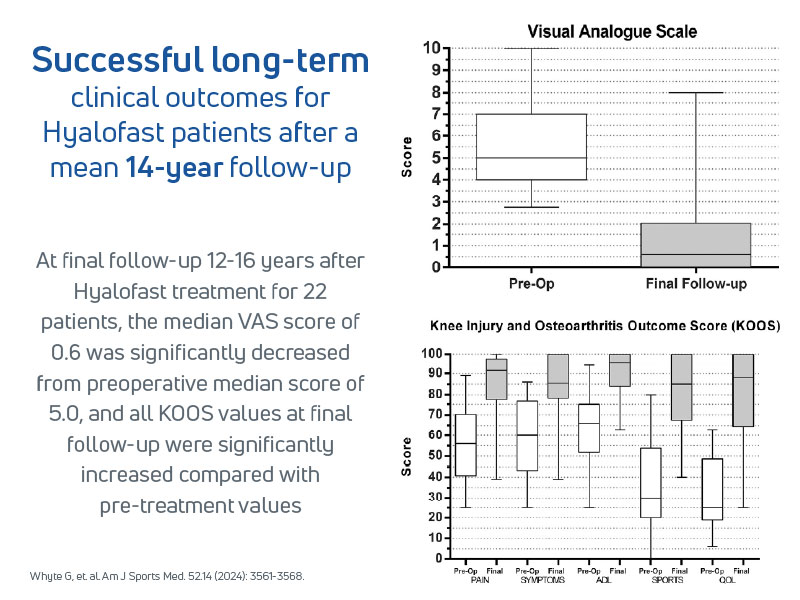
- 30 patients with full-thickness knee lesions, treated with Hyalofast and BMAC, were followed for a mean of 14 years
- Sustained improvements in pain, function, and activity scores over time
- MRI confirmed durable cartilage repair and integration
- Low reoperation rate and high patient satisfaction
(Hakan Sofu , Nizamettin Kockara , Ali Oner , Yalkin Camurcu , Ahmet Issın , Vedat Sahin )
Key Takeaways
- 48 patients treated with microfracture alone or with Hyalofast applied after microfracture
- Scaffold group had better clinical scores and MRI outcomes at 24 months
- No major complications reported
- Supports scaffold use as a more effective one-step treatment
(Alberto Gobbi, Celeste Scotti, Georgios Karnatzikos, Abhishek Mudhigere, Marc Castro, Giuseppe M. Peretti)
Key Takeaways
- 30 patients aged 45+ with full-thickness knee lesions treated with Hyalofast and BMAC
- Significant improvements in clinical scores at 12 and 24 months
- MRI showed cartilage maturation and integration
- Safe and effective option for older patients
Resources
Product Information
Access comprehensive technical documentation and marketing materials
- Brochures
- Rehab Guidelines
Patient Resources
Educational materials and patient success stories
- Patient Testimonial
- Blog
Videos
Animation and educational video content
- Surgical Technique Videos
- Hyalofast Overview
- Patient Testimonial Video
Instructions for Use
Complete product specifications and safety information
Hyalofast is CE-marked approved as a biodegradable support for the entrapment of mesenchymal stem cells for the repair of chondral or osteochondral lesions. Hyalofast may act as a support for bone marrow aspirate concentrate or as a chondroprotective coverage which favors in situ residence of mesenchymal stem cells after their mobilization due to microfracture or perforation procedures.
Hyalofast is not yet available in the United States, but important progress is underway toward U.S. FDA approval. Anika is actively engaged in ongoing dialogue with the FDA while under Premarket Approval (PMA) review. This regulatory pathway reflects Anika’s commitment to making Hyalofast accessible to U.S. patients. Already used internationally for cartilage repair, Hyalofast has shown promising clinical safety and efficacy.
- Pasquinelli G, Orrico C, Foroni L, Bonafè F, Carboni M, Guarnieri C, Raimondo S, Penna C, Geuna S, Pagliaro P, Freyrie A, Stella A, Caldarera CM, Muscari C. Mesenchymal stem cell interaction with a non-woven hyaluronan-based scaffold suitable for tissue repair. J Anat. 2008 Nov;213(5):520-30. https://pubmed.ncbi.nlm.nih.gov/19014359/(opens in a new tab)
- https://www.anikaifu.com/wp-content/uploads/2024/12/AML-500-323_Rev-E.pdf(opens in a new tab)
- Cavallo C, Desando G, Columbaro M, Ferrari A, Zini N, Facchini A, Grigolo B. 2012. Chondrogenic differentiation of bone marrow concentrate grown onto a hylauronan scaffold: Rationale for its use in the treatment of cartilage lesions. J Biomed Mater Res Part A 2012:00A:000–000. https://onlinelibrary.wiley.com/doi/10.1002/jbm.a.34460(opens in a new tab)
- Facchini, Andrea & Lisignoli, Gina & Cristino, Sandra & Roseti, Livia & Franceschi, Luciana & Marconi, Emanuele & Grigolo, Brunella. (2006). Human chondrocytes and mesenchymal stem cells grown onto engineered scaffold. Biorheology. 43. 471-80. https://pubmed.ncbi.nlm.nih.gov/16912418/
- Lee, Y. H. D., Suzer, F., & Thermann, H. (2014). Autologous Matrix-Induced Chondrogenesis in the Knee: A Review. Cartilage, 5(3), 145–153. https://journals.sagepub.com/doi/pdf/10.1177/1947603514529445?download=true(opens in a new tab)
- Implantation Study of a Femoral Osteochondral Cylindrical Defect in Rabbits – NAMSA – Anika Data on file
- Buda R, et al. Osteochondral lesions of the knee: a new one-step repair technique with bone-marrow-derived cells. J Bone Joint Surg Am. 2010 Dec. https://pubmed.ncbi.nlm.nih.gov/21123588/(opens in a new tab)
- Buda R. et al. One-step arthroscopic technique for the treatment of osteochondral lesions of the knee with bone-marrow-derived cells: three years results. Musculoskelet Surg. 2013 Feb. https://pubmed.ncbi.nlm.nih.gov/23420394/(opens in a new tab)
- Vannini F. et al. One Step Treatment of Juvenile Osteochondritis Dissecans in the Knee: clinical results and T2 mapping Characterization. Orthop Clin North Am, 2012. https://pubmed.ncbi.nlm.nih.gov/22480472/(opens in a new tab)
- Gobbi A. et al. Matrix-Induced Autologous Chondrocyte Implantation versus Multipotent Stem Cells for the treatment of large patellofemoral chondral lesions: a non randomized prospective trial. Cartilage 2014 Dec. https://pmc.ncbi.nlm.nih.gov/articles/PMC4462249/pdf/10.1177_1947603514563597.pdf(opens in a new tab)
- Gobbi A., et al. One-step surgery with multipotent stem cells and Hyaluronan-based scaffold for the treatment of full-thickness chondral defects of the knee in patients older than 45 years. Knee Surg Sports Traumatol Arthrosc, 2017. https://pmc.ncbi.nlm.nih.gov/articles/PMC5548854/pdf/167_2016_Article_3984.pdf(opens in a new tab)
- Whyte GP, Bizzoco L, Gobbi A. One-Step Cartilage Repair of Full-Thickness Knee Chondral Lesions Using a Hyaluronic Acid-Based Scaffold Embedded With Bone Marrow Aspirate Concentrate: Long-term Outcomes After Mean Follow-up Duration of 14 Years. Am J Sports Med. 2024 Dec;52(14):3561-3568.Epub 2024 Nov 3. https://pubmed.ncbi.nlm.nih.gov/39491512/(opens in a new tab)
- Gobbi A, Whyte GP. One-Stage Cartilage Repair Using a Hyaluronic Acid-Based Scaffold With Activated Bone Marrow-Derived Mesenchymal Stem Cells Compared With Microfracture: Five-Year Follow-up. Am J Sports Med. 2016 Nov;44(11):2846-2854. Epub 2016 Jul 29. https://pubmed.ncbi.nlm.nih.gov/27474386/(opens in a new tab)
- Sofu H, Kockara N, Oner A, Camurcu Y, Issın A, Sahin V. Results of Hyaluronic Acid-Based Cell-Free Scaffold Application in Combination With Microfracture for the Treatment of Osteochondral Lesions of the Knee: 2-Year Comparative Study. Arthroscopy. 2017 Jan;33(1):209-216. Epub 2016 Sep 7. https://pubmed.ncbi.nlm.nih.gov/27614391/(opens in a new tab)

