What Is Tactoset?
Tactoset is a synthetic, biocompatible, calcium phosphate bone graft substitute enhanced with hyaluronic acid (HA), a substance naturally found throughout the human body. It is highly flowable and easily injectable during minimally invasive surgery and is designed to fill insufficiency fractures, reinforce bone structure, and augment hardware such as suture anchors and other hardware. During the healing process, Tactoset supports new bone growth, helping people return to activity with minimal surgical intervention.
Once injected, Tactoset hardens quickly and mimics the properties of trabecular bone. It is osteoconductive and supports cell-mediated regeneration of new bone as it is resorbed and replaced by the growth of new bone during the healing process.1,2
Supporting a variety of uses, Tactoset is indicated for:
- Filling bone voids, bone cysts or defects of the skeletal system (may also be combined with autologous bone marrow aspirate (BMA))2
- Augmenting hardware, such as suture anchors and screws2
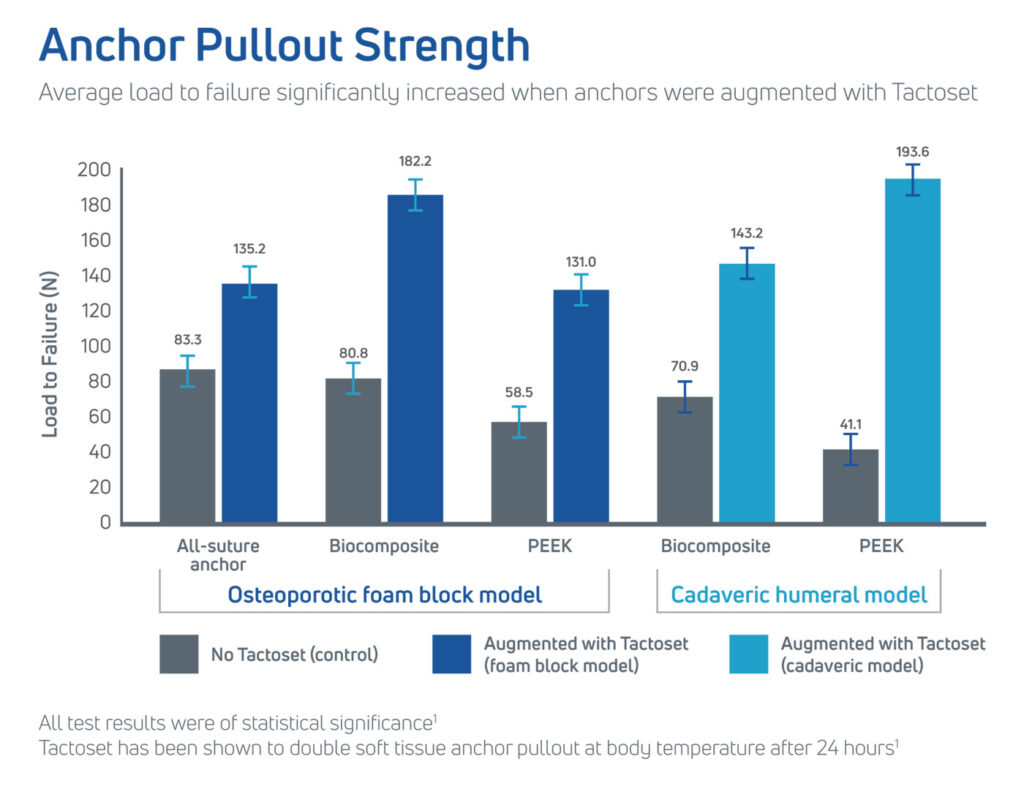
Using Tactoset to augment hardware increases fixation strength and the density of poor-quality bone caused by cysts, osteoporosis, or osteoarthritis. When using Tactoset with suture anchors, it allows the hardware to be placed securely in the ideal repair location and can achieve greater than twice the pullout strength of non-augmented anchors.1,3
Real Life Advantages
Formulation & Core Properties
- Proprietary hyaluronic acid-enhanced formulation
- Injectable, self-setting, osteoconductive bone void filler
- Highly flowable and easily injectable during minimally invasive surgery1,2
- Interdigitates into trabecular bone architecture to fill closed bone voids1,4
- Closed mixing system minimizes environmental exposure1
- Supports endogenous cell-mediated regeneration of new bone as material is resorbed1,2
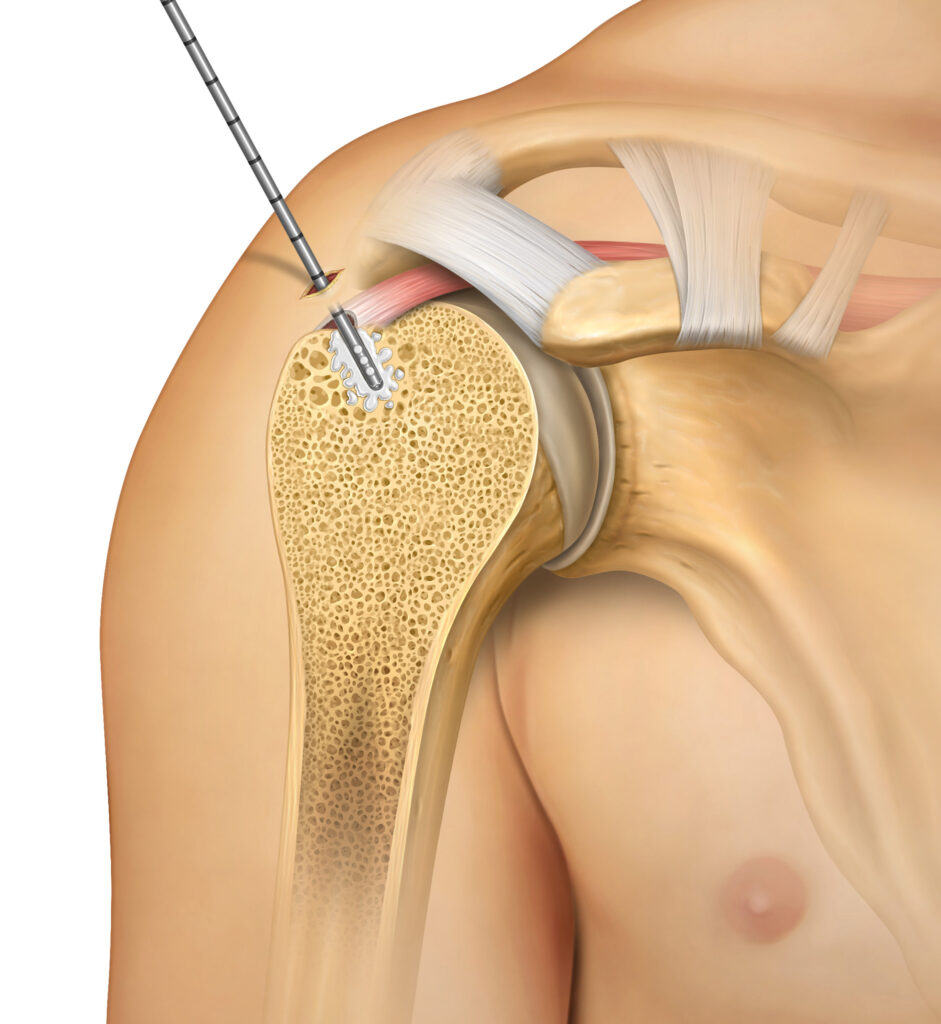
Hardware Augmentation Indication
- Can augment hardware, such as suture anchors and screws
- Provides greater than twice the pullout strength of non-augmented anchors1,3

Combination with Bone Marrow Aspirate (BMA)
- Can be combined with BMA, leveraging a patient’s own biology2
- Available supporting Tactoset’s BMA indication, the Marrow Cellution™ bone marrow aspiration needle extracts high-quality, high-cell-count marrow from multiple geographies while limiting peripheral blood dilution and eliminating the need for centrifugation
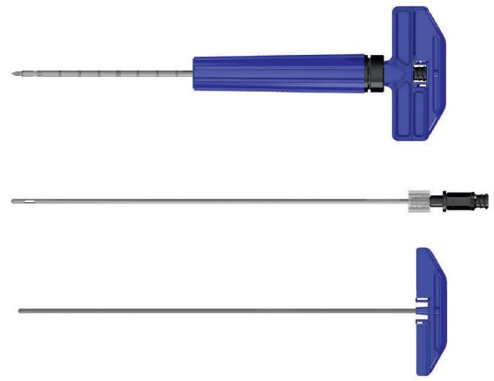
Surgical Handling & Procedural Advantages
- Unique cannula design allows surgeons to easily switch intraoperatively between side and end Tactoset delivery without having to remove and reinsert the outer cannula1
- Can be performed as an outpatient procedure in an ambulatory surgical center (ASC)

Evidence
Tactoset improves the structural quality of subchondral bone, increases pullout strength of soft tissue anchors, and supports regeneration of new bone during the healing process.
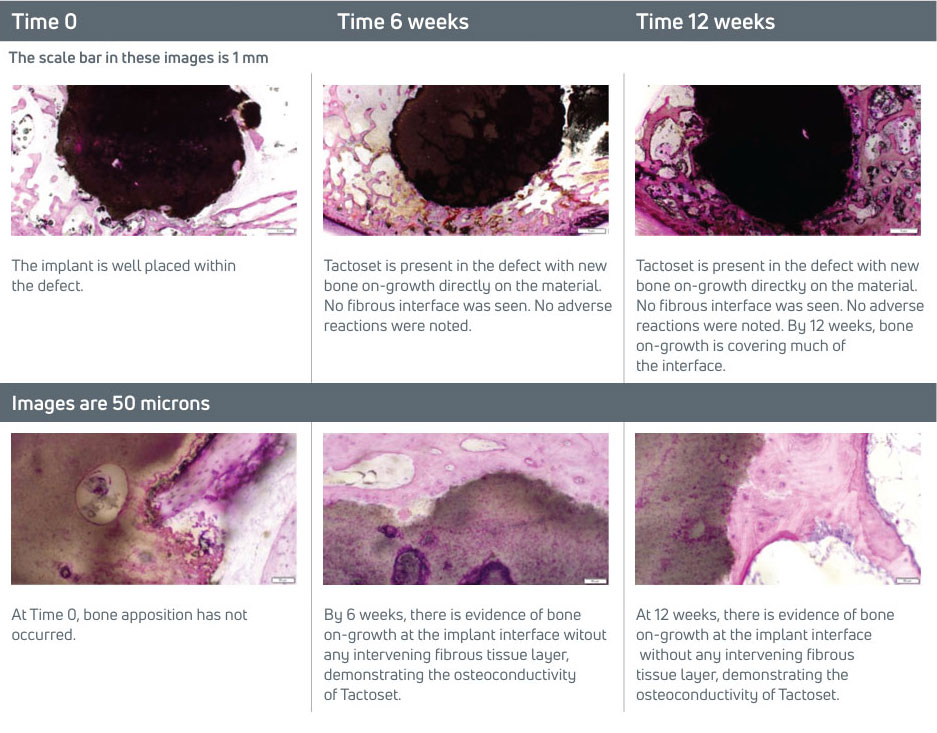
In a pre-clinical study, the in vivo performance of Tactoset was evaluated in cancellous bone defects.
Select Publications & Clinical Data
(Miguel A. Diaz, Steven Munassi, David E. Teytelbaum, Anthony Pipitone, Christopher E. Baker)
Key Takeaways
- The purpose of the study was to compare various suture anchor designs with and without calcium phosphate (CaP) augmentation in an osteoporotic foam block model and decorticated proximal humerus cadaveric model
- Augmenting various suture anchors with CaP has shown to significantly increase pull-out strength and stiffness in an osteoporotic foam block and time zero cadaveric bone model
(Dennis DeBernardis, Michael Stark, Elizabeth Ford, Christopher McDowell, Sean McMillan)
Key Takeaways
- Retrospective single-surgeon analysis of 74 patients with painful subchondral bone marrow edema (SBME) of the knee confirmed on MRI
- Patients undergoing percutaneous skeletal fixation (PSF) for the treatment of painful SBME of the knee experienced an overall success rate of 82.4% in prevention of additional intervention within a 2-year postoperative period
(Michael Stark, Dennis DeBernardis, Chris McDowell, Elizabeth Ford, Sean McMillan)
Key Takeaways
- This paper presents percutaneous skeletal fixation (PSF) technique utilizing Tactoset, an injectable synthetic, biocompatible hyaluronic acid-based bone graft substitute that allows for minimally invasive treatment for painful subchondral bone marrow edema (SBME)
- Through appropriate patient identification, proper techniques, and the management of patient expectations, the Tactoset system can allow for the reduction of pain in patients with painful SBME in a minimally invasive way
Resources
Product Information
Access comprehensive technical documentation and marketing materials
- Brochure
- Tech Guides
Patient Resources
Educational materials and patient success stories
- Patient Brochure
- Blog
Instructions for Use
Complete product specifications and safety information
Tactoset is available in the United States (US) and select markets outside of the US and is indicated for filling bone voids or defects of the skeletal system that are not intrinsic to the stability of the bony structure. Tactoset can augment hardware and support bone fragments during the surgical procedure. The cured paste acts only as a temporary support media and is not intended to provide structural support during the healing process.
Anika products may not be available in all geographies. Product availability is subject to the regulatory clearances in individual markets. Please reach out to your local representative or Contact Us if you have questions about specific market approvals
For complete product information, including indications, contraindications, warnings, and precautions, please refer to the Instructions for Use found here(opens in a new tab).
- Data on file, Anika Therapeutics, Inc.
- Tactoset Instructions for Use. AML 500-335.
- Diaz M, Munassi S, Teytelbaum D, Pipitone A, Baker C. An Injectable Calcium Phosphate Bone Graft Substitute Improves the Pullout Strength of Various Suture Anchor Designs in an Osteoporotic Bone Model. Arthroscopy, Sports Medicine, and Rehabilitation 2023 Jan 5; pp e1-e11. doi:10.1016/j.asmr.2023.01.010
- Stark M, DeBernardis D, McDowell C, Ford E, McMillan S. Percutaneous Skeletal Fixation of Painful Subchondral Bone Marrow Edema Utilizing an Injectable, Synthetic, Biocompatible Hyaluronic Acid-Based Bone Graft Substitute. Arthroscopy Techniques 2020 Nov; Vol 9, No 11; pp e1645-e1650. doi:10.1016/j.eats.2020.07.005