What Is Integrity?
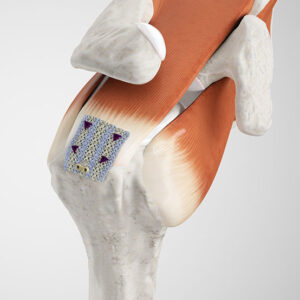
The Integrity™ Implant is a hyaluronic acid (HA)-based scaffold for tendon repair that provides reliable strength and regenerative biology. The implant is a porous, knitted scaffold constructed from Hyaff®, our proprietary HA fiber, that supports tissue regeneration, resorbs over time, and is reinforced with PET (polyethylene terephthalate), a permanent material commonly used in sutures that is well known for its biostability and promotion of tissue ingrowth.1 By utilizing both Hyaff and PET, Integrity is a unique hybrid structure that offers biologic enhancement and strength for confident implantation.
Integrity is available in multiple shapes and sizes, 20x25mm, 25x30mm, 25x60mm, and 40x60mm, all of which are 80% Hyaff and 20% PET. The wide range of available sizes offers intra-operative flexibility, supporting a broad spectrum of tendon repair procedures in the shoulder, knee, hip, foot, and ankle.
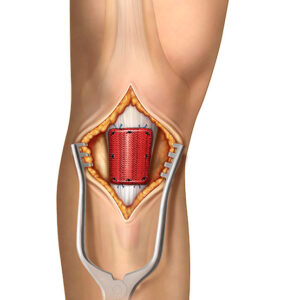
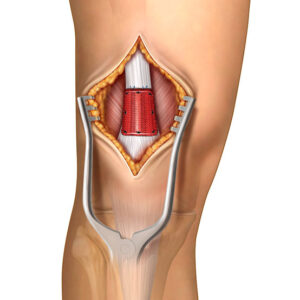
Real Life Advantages
Tendon augmentation is designed to support regenerative healing and thickening of tendons for a strong recovery. Integrity promotes tendon regeneration by incorporating hyaluronic acid, which enhances the body’s cellular response and supports the natural healing process.
Reliable strength
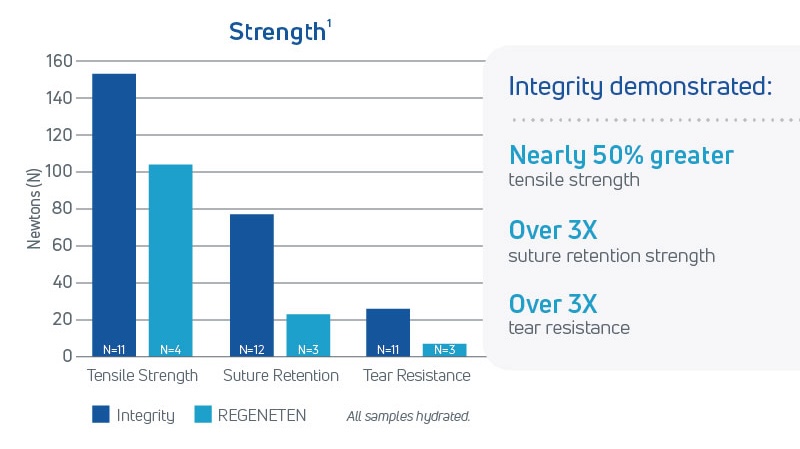
- Integrity provides higher tensile strength, suture retention, and tear resistance in a thin knitted format, even when wet2
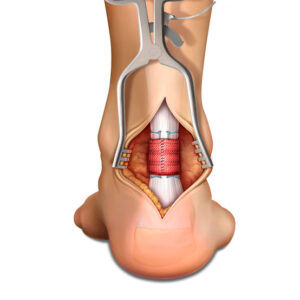
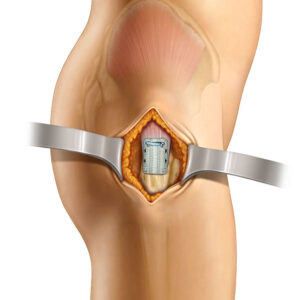
- Inherently strong scaffold can be confidently manipulated arthroscopically and enables surgical versatility
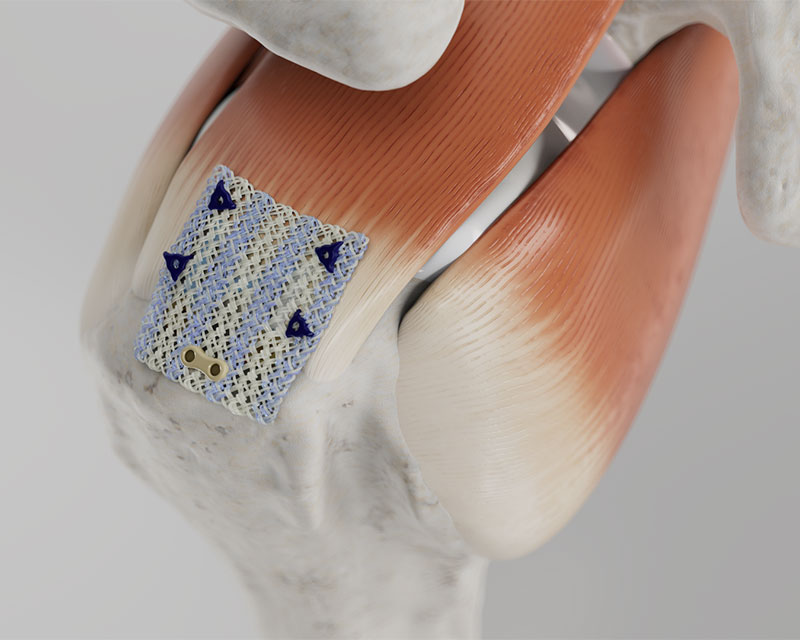
Streamlined technique for rotator cuff repair
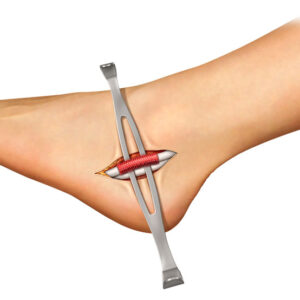
- “Lateral first” fixation, unique instrumentation, and simplified surgical technique allow for precise implant placement
- Delivery instruments are single use and provided sterile for added efficiency
Evidence
In a head-to-head pre-clinical study comparing Anika’s Integrity Implant and REGENETEN®, results showed:
- As early as 12 weeks post-implantation, fibroblast infiltration and regularly oriented new collagenous tissue formation had occurred within the Integrity repair, demonstrating greater regenerative capacity compared to REGENETEN3
- At 26 weeks, within the resorbing Integrity structure, new collagenous tissue infiltration forming a new network of tendon tissue had occurred, resulting in nearly 3 times greater thickness in the repaired tendon than REGENETEN3
- Increased tendon thickness is thought to improve the local biomechanical environment of the tear by reducing tendon strain, thus optimizing its healing potential6
Clinical Highlights: Integrity Implant System for Rotator Cuff Repair7
A recent white paper evaluating 29 patients treated with the Integrity Implant System for partial and full thickness rotator cuff tears revealed compelling early outcomes:
- Pain relief and functional improvement sustained over 6 months7
- No device-related complications reported7
- No evidence of retears at the 6-month follow-up7
The Integrity Implant System shows promise as a biologic strategy to support tendon healing in rotator cuff repair.2,3,7,8
Resources
Product Information
Access comprehensive technical documentation and marketing materials
- Brochures
- Technique Guides
- White Papers
Patient Resources
Educational materials and patient success stories
- Patient Testimonial Videos
- Patient Brochure
- Blog
Videos
Animation and educational video content
- Integrity Mode of Action animation
- Patient Testimonials
- Surgical Technique Videos
Instructions for Use
Complete product specifications and safety information
The Integrity Implant System is available in the US and select markets outside of the United States and is indicated for the management and protection of tendon injuries in which there has been no substantial loss of tendon tissue.
Anika products may not be available in all geographies. Product availability is subject to the regulatory clearances in individual markets. Please reach out to your local representative or Contact Us if you have questions about specific market approvals.
For complete product information, including indications, contraindications, warnings, and precautions, please refer to the Instructions for Use found here(opens in a new tab).
REGENETEN is a registered trademark of Smith+Nephew.
- Metzger, Anja. “Polyethylene terephthalate and the pillar™ palatal implant: its historical usage and durability in medical applications.” Biomedical Engineering 11.9 (1976): 301-306
- Data on file, Anika Therapeutics, Inc.
- Walsh, WR et al. “Rotator Cuff Healing with Anika Technology” Data on File, Anika Therapeutics, Inc.
- Hu, Min, et al. “Three dimensional hyaluronic acid grafts promote healing and reduce scar formation in skin incision wounds.” J. Biomed. Mater. Res., 67.1 (2003): 586-592
- Vindigni, V. Hyaluronan Benzyl Ester as a Scaffold for Tissue Engineering. Int. J. Mol. Sci. 2009, 10, 2972-2985; doi:10.3390/ijms10072972
- Schlegal, T. F., M.D. (2017). Radiologic and clinical evaluation of a bioabsorbable collagen implant to treat partial-thickness tears: A prospective multicenter study. Journal of Shoulder and Elbow Surgery. https://doi.org/10.1016/j.jse.2017.08.023(opens in a new tab)
- Porter, David A., Pietrzak, William S. The Integrity Implant System: Early Outcomes Following Rotator Cuff Repair. (2025) White Paper, Anika Therapeutics, Inc. AML-900-735 REV 01.
- Patel, Raahil, et al. “Hyaluronic Acid− Biocompatible Implant System for Rotator Cuff Augmentation.” Arthroscopy Techniques (2025): 103990.



