What Is Orthovisc?
Orthovisc® is a sterile, ultra-pure hyaluronic acid (HA) injection developed to relieve knee pain associated with osteoarthritis (OA). It consists of high molecular weight sodium hyaluronate derived from bacterial fermentation, suspended in a physiologic saline solution.1
HA is a naturally occurring substance in synovial fluid that provides essential lubrication and shock absorption within the joint.
In OA, the quality and quantity of natural HA diminish, leading to joint pain and stiffness. Orthovisc supplements this loss by restoring the viscoelastic properties of the synovial fluid, helping to reduce friction and improve joint function.1
Orthovisc is FDA approved for use in the knee and is typically administered as a series of 3 or 4 intra-articular injections, given at weekly intervals.1 In the European Economic Area and other parts of the world, Orthovisc is indicated as a viscoelastic supplement or a replacement for synovial fluid in human joints. It is intended for patients who have not responded adequately to conservative treatments such as physical therapy or over-the-counter anti-inflammatory pain relievers (NSAIDs) such as acetaminophen or ibuprofen.1
Real Life Advantages
- Long-lasting pain relief: HA provides long-lasting pain relief up to 6 months2
- Natural solution: Highly concentrated, non-animal-based HA produced from bacterial fermentation
- High HA concentration: 15 mg/mL, one of the highest available in its class
- Biocompatible and resorbable: Safely and naturally broken down by the body3,4,5
- Excellent safety profile: No confirmed pseudoseptic or severe inflammatory reactions2,5
- Multi-injection regimen: Simple procedure typically administered during a routine office visit once a week for 3 or 4 weeks1
- Proven patient outcomes: Significant improvements in pain, stiffness, and physical function2
The Science Behind Orthovisc
Orthovisc mimics the natural hyaluronic acid found in healthy synovial fluid. Its high molecular weight and concentration enhance the viscoelastic properties of the joint fluid, improving its ability to cushion and lubricate the joint.
The hyaluronic acid used in Orthovisc is derived from bacterial fermentation, ensuring high purity, biocompatibility, and eliminating the risk of animal-derived contaminants. This makes it suitable for a wide range of patients, including those with sensitivities or allergies to animal proteins.
How It Works
All joints in the body contain synovial fluid, a vital substance that provides lubrication, cushioning, and protection as we move. This fluid is naturally rich in hyaluronic acid (HA), which helps reduce friction and absorb shock during joint activity.
Over time, especially in patients with osteoarthritis (OA), the synovial fluid becomes degraded, losing both its quality and volume. This leads to increased joint friction, inflammation, stiffness, and pain.
Orthovisc is a high molecular weight HA injection designed to supplement the deteriorated synovial fluid in osteoarthritic joints, particularly the knee. Derived from bacterial fermentation, Orthovisc offers high purity and excellent biocompatibility, making it a safe and effective option for many patients.
Once injected directly into the joint space, Orthovisc helps to:
-
- Restore lubrication and cushioning
- Reduce inflammation and joint friction
- Protect cartilage from further wear and tear
This results in improved mobility and pain relief, with effects that can last for up to six months or longer. Orthovisc is especially beneficial for patients who have not responded to conservative treatments and are seeking to delay or avoid surgical intervention. Its multi-injection regimen allows for tailored dosing over several weeks, offering sustained relief and flexibility in treatment planning.
Evidence
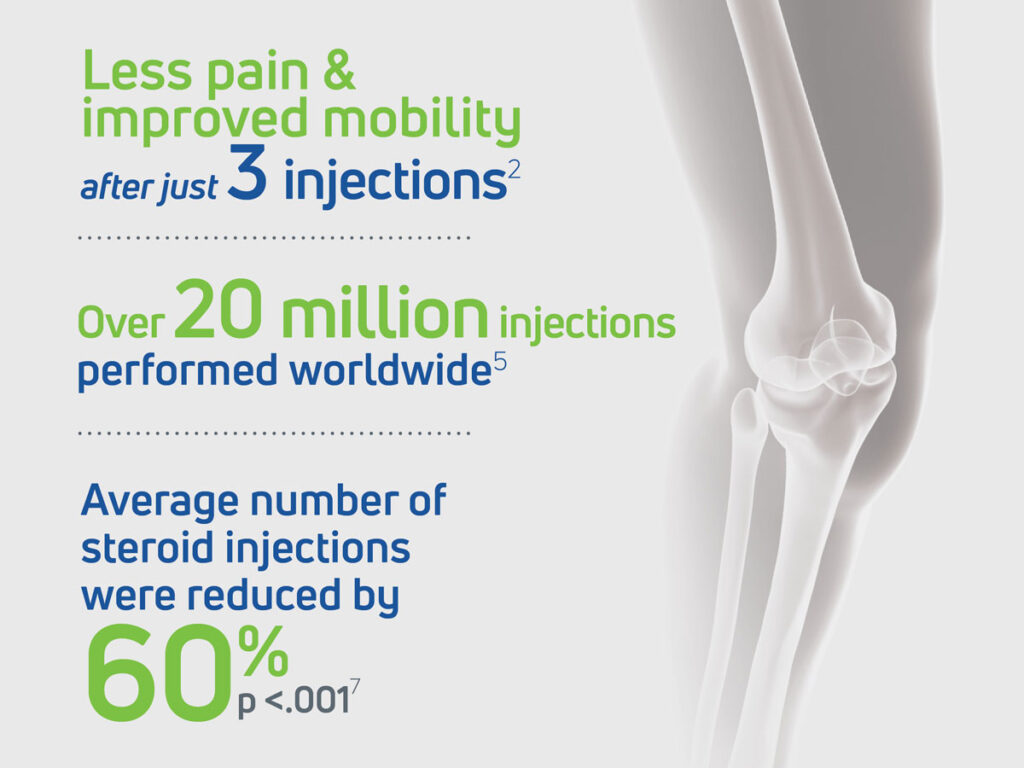
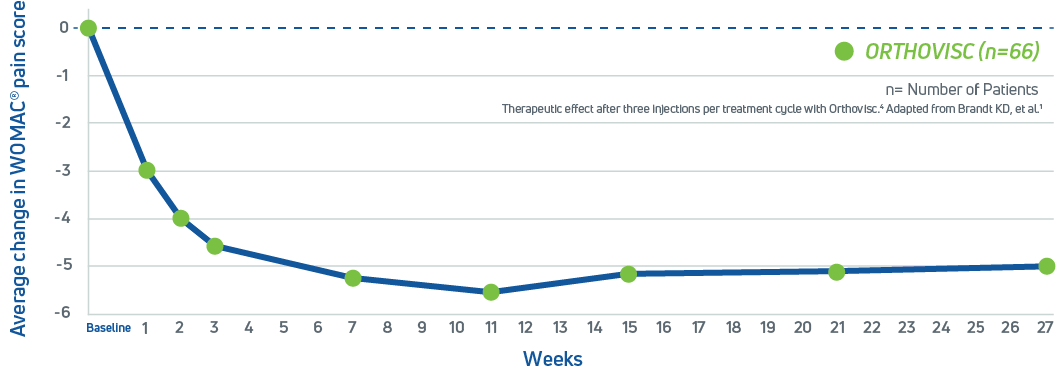
Orthovisc has been evaluated in several U.S. clinical trials, including a study of the effectiveness of sodium hyaluronate in the treatment of knee osteoarthritis which found that after just three intra-articular injections of Orthovisc spaced one week apart, patients experienced a significant reduction in joint pain and improved mobility over a period of 27 weeks2
A pivotal randomized, controlled, multicenter study published in The Journal of Rheumatology, which demonstrated statistically significant improvements in pain relief and joint function.6 In a 6-month follow-up, patients reported sustained benefits in pain reduction and quality of life.
A retrospective study of nearly 30,000 patients demonstrates that Orthovisc offers effective pain alleviation among knee OA patients while reducing the use of prescription pain medications such as steroids, NSAIDs (non-steroidal anti-inflammatory drugs) such as Ibuprofen or Aspirin, and opioids.7
Since its launch, over 21 million Orthovisc injections have been performed worldwide.5 This reflects its widespread adoption and clinical utility in managing osteoarthritis pain, particularly in the knee.


Select Publications & Clinical Data
(Abhishek S. Chitnis, Katherine Etter, Chantal E. Holy, F. Scott Gray, Fernando J. Manalac, Brad Bisson, Samir K. Bhattacharyya)
Key Takeaways
- Retrospective real-world analysis evaluating the effect of high-concentration, non-avian hyaluronic acid (HA) on pain medication use in knee osteoarthritis patients
- Significant reduction in opioid and corticosteroid use observed following HA treatment
- Patients showed a reduction in pain-medication prescription fills post-treatment, indicating improved symptom control
- The study supports the clinical utility of HA injections in reducing reliance on pharmacologic pain management, especially opioids
- Conclusion: High molecular weight HA may offer effective symptom relief in OA while contributing to safer, more sustainable pain management strategies
(Katherine Etter, Abhishek S. Chitnis, Chantal E. Holy, F. Scott Gray, Fernando J. Manalac, Brad Bisson, Samir K. Bhattacharyya)
Key Takeaways
- Retrospective real-world study assessing whether high-concentration non-avian high molecular weight hyaluronic acid (HA) injections delay time-to-total knee replacement (TKR) in osteoarthritis patients
- Patients receiving HA injections had a longer time-to-TKR compared to those who did not receive HA treatment
- The study suggests that viscosupplementation with high-concentration HA may postpone surgical intervention, offering extended symptom management
- Supports the role of HA injections as a non-surgical option that may reduce healthcare costs and improve patient quality of life by delaying joint replacement
- Conclusion: High-concentration non-avian HA injections may be an effective strategy to defer TKR in OA patients, especially those seeking to avoid or delay surgery
Resources
Product Information
Access comprehensive technical documentation and marketing materials
- Brochure
Instructions for Use
Complete product specifications and safety information
Looking for Orthovisc in the US?
In the United States, Orthovisc and Monovisc are exclusively distributed by J&J MedTech.

To learn more about these products or connect with a representative, please visit the J&J MedTech website(opens in a new tab).
In the U.S., Orthovisc is indicated in the treatment of osteoarthritis (OA) of the knee in patients who have failed to respond adequately to conservative non-pharmacologic therapy and to simple analgesics, e.g. acetaminophen.1
In Canada, Orthovisc is indicated for the symptomatic treatment of osteoarthritis of the knee.8
In the European Economic Area, Orthovisc is indicated as a viscoelastic supplement or a replacement for synovial fluid in human joints. Orthovisc is well suited to provide relief for the pain of osteoarthritis. The actions of Orthovisc are lubrication and mechanical support.9,10
Anika products may not be available in all geographies. Product availability is subject to the regulatory clearances in individual markets. Please reach out to your local representative or Contact Us if you have questions about specific market approvals.
Important Safety Information
ORTHOVISC should not be taken if you are allergic to hyaluronate products. If you have any known allergies, you should consult with your healthcare professional. You should not have an injection into the knee if you have infections or skin diseases around the injection site. Possible side effects of ORTHOVISC injections in the knee joint: pain, swelling, heat, rash, itching, bruising and/or redness. You may also feel achy. These reactions are generally mild and do not last long. If any of these symptoms or signs appear after you are given ORTHOVISC or if you have any other problems, you should call your healthcare professional.
For complete product information, including indications, contraindications, warnings, and precautions, please refer to the Instructions for Use found here(opens in a new tab).
- AML 500-254 REV D 2023-07 IFU
- Brandt KD, Block JA, Michalski JP, et al. Efficacy and safety of intra-articular sodium hyaluronate in knee osteoarthritis. ORTHOVISC Study Group. Clin OrthopRelat Res 2001;(385):130–43. https://europepmc.org/article/med/11302304(opens in a new tab)
- Richter, W., (1974) Non-immunogenicity of a Purified Hyaluronic Acid Preparation tested by Passive Cutaneous Anaphylaxis. Int. Arc. Allergy 47,211.
- Richter, W., Ryde, E.M. and Zetterstrom, E.O. (1979) Non-immunogenicity of a Purified Sodium Hyaluronate Preparation in Man. Int. Arch. Appl. Immunol. 59,455.
- Data on file, Anika Therapeutics, Inc.
- Neustadt D, Caldwell J, Bell M, Wade J, and Gimbel J. Clinical effects of intraarticular injection of high molecular weight hyaluronan (ORTHOVISC) in osteoarthritis of the knee: A randomized, controlled, multicenter trial. J. Rheumatology 2005; (32): 1928-36. https://www.jrheum.org/content/jrheum/32/10/1928.full.pdf(opens in a new tab)
- Abhishek S. Chitnis, Katherine Etter, Chantal E. Holy, F. Scott Gray, Fernando J. Manalac, Brad Bisson & Samir K. Bhattacharyya (2019): Real-world impact of the high concentration non-avian high molecular weight hyaluronan on pain medication use among osteoarthritis patients. Curr Med Res Opin 2019.
DOI: 10.1080/03007995.2019.1596670 https://www.tandfonline.com/doi/10.1080/03007995.2019.1596670?url_ver=Z39.88-2003&rfr_id=ori:rid:crossref.org&rfr_dat=cr_pub%20%200pubmed(opens in a new tab) - AML 500-269/B IFU
- AML 500-261 REV-F IFU
- AML 500-271 REV-F IFU